en
Medical Intertrade d.o.o. is a company registered and authorized by the Agency for medicinal products and medical devices for foreign trade, wholesale and retail trade of non-alimentary products, whose core business is trade in medicinal products, medical devices and medical equipment.
Overall business activities are harmonized with EU Directives and Croatian legislation, respecting the principles of good warehousing practice and good distribution practice of pharmaceutical products, medical devices and equipment.
All activities that influence the quality of the product are stipulated by Standard Operating Procedures (SOP) and Work Instructions (WI) that are constituent parts of an integrated Quality and Environmental Management System in line with international norms ISO 9001:2015 and ISO 14001:2015. Control of the efficiency of implementation of proscribed procedures is regularly conducted by the global certification organization and by our many partners.
Well chosen business strategy gives us the energy and enthusiasm for the realization of new business challenges.
Integrated management system is the integral part of our approach for constant improvements that are reflected in all areas of company activities and that are managed by educated and expert employees. All segments of company activities have fully ensured requirements for handling of products, warehousing and transport in controlled conditions, including cold chain and identification and traceability of every product per batch number and expiry date.
Founding values that drive our business, high organizational level and excellence in every segment, have been recognized by our customers and suppliers.

Agency contracts
Membership

Our achievements
delivery points
in terms of total turnover of all companies in Croatia
largest company in the wholesale sector in Croatia
years of existence

Mission & vision
MISSION
VISION
We are one of the leading wholesalers of medicinal products and medical devices in the Republic of Croatia and have an important place among the participants in the health care system. With a wide range of products, many years of experience and a unique approach, we ensure the best logistics solutions and professional support to business partners. We are dedicated to corporate social responsibility, creating a stimulating working environment as well as taking care of environmental protection.
As quality of processes and services are imperative in our activities, we have implemented quality management systems according to ISO standards. Continuous improvements of process and service implementation have been confirmed by successful re-certification by regular audits.
Certificate of GDP Compliance of a Wholesale Distributor
Certificate of GDP Compliance of a Wholesale Distributor - Gospodarska Ulica 2
Certificate of GMP Compliance of a Manufacturer
ISO 9001:2015
ISO 14001:2015
History
Locations
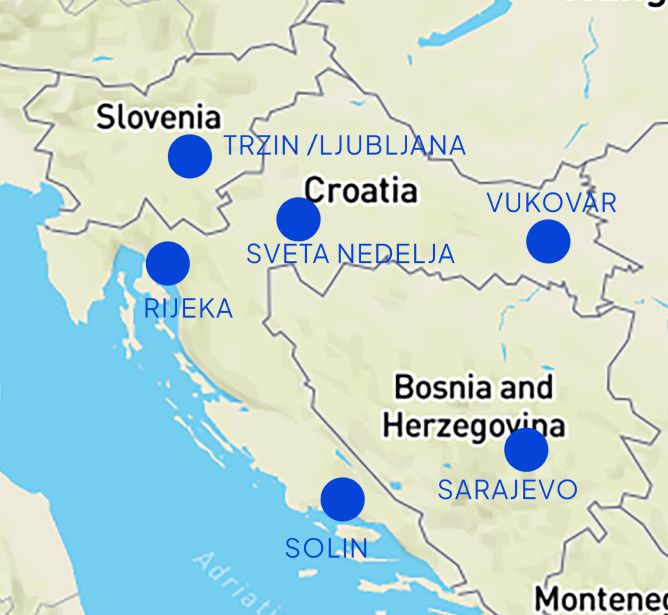
Sveta Nedelja
Vukovar
Dugopolje
Rijeka
Trzin / Ljubljana
Sarajevo





















